Alkaline industrial wastewater and CO2 – the perfect pairing?
Alkaline industrial wastewater is commonly neutralized with acid like sulfuric or hypochlorite acid. Purchasing, storing, and dosing the acid involves costs. Instead, scientists at the Helmholtz Centre Hereon believe that all that is needed is to introduce CO2 into the water. They have developed a market-ready process that combines the treatment of alkaline wastewater with CO2 emission reduction.
Making effective use of a well-known phenomenon
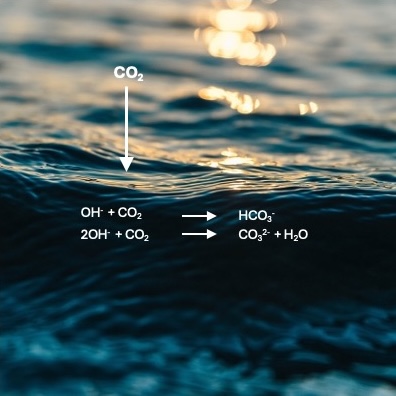
When CO₂ dissolves in alkaline water, it forms carbonic acid, which reacts with the hydroxide ions present to form bicarbonate. In this form, the greenhouse gas can remain bound in the water for long periods and be transported to the ocean via rivers. In nature, bicarbonates are formed when CO2 binds to carbonates, which enter rivers and oceans through the weathering of limestone.
For some time now, researchers have been considering the targeted injection of carbonates into water bodies to increase their CO2 absorption capacity. The team led by Prof. Hemuth Thomas, head of the Hereon Institute of Carbon Cycles, has now developed an industrial-scale technical process that can neutralize alkaline industrial wastewater by introducing CO2. This process not only saves large quantities of mineral acids but will also enable the sequestration of many millions of tons of CO2 per year in the future.
The process described in the journal Environmental Science & Technology Letters does not involve complete neutralization, as achieved by adding acid, but rather the conversion of hydroxide alkalinity into carbonate alkalinity. The authors refer to this as Wastewater Alkalinity Preservation (WAP).
Why industrial wastewater?
In certain industrial facilities, such as those involved in aluminum and cement production, the manufacturing process generates both large volumes of alkaline wastewater and significant amounts of CO2, the release of which into the atmosphere is to be prevented in the future. The volumes of wastewater and CO2 at such a site are generally known.
By taking into account the alkalinity or carbonate content of the receiving water body, the CO2 dosage in the wastewater or its dilution can be adjusted so that no CO2 is released when the treated wastewater is discharged and environmental protection requirements — particularly the pH value — for the discharge are met.
For certain industrial facilities, Helmuth Thomas calculated the expected carbon dioxide turnover and the potential for reducing emissions between 2030 and 2100. Assuming a relatively small increase in the global volume of alkaline wastewater streams (1.18% per year), the potential for CO2 emission reductions during this period could rise from approximately 30 million tons per year to about 50 million tons per year. With greater global economic growth, wastewater volumes and, accordingly, CO2 absorption capacities would also increase (as estimated from a graph published in the study).
Technical implementation and monitoring
In addition to simulating the potential for sequestering CO2 using alkaline wastewater, a plant design was also developed. The purpose of this design is, on the one hand, to ensure that the injected CO2 dissolves completely and, on the other hand, to enable the precise quantification of CO2 uptake.
Contact between the wastewater and the gas is established in a static mixer, which requires energy only for startup and is then maintained in continuous operation by hydraulic suction.
The entire system is equipped with sensors to measure the pH of the wastewater, the gas flow rate, and the CO2 partial pressures and pH values in the receiving water body upstream and downstream of the gas-wastewater mixing chamber.
According to the authors, the entire process is based on proven components that are commercially available. Therefore it corresponds to TRL (Technology Readiness Level) 9, meaning it can be implemented on an industrial scale immediately.


