New method for eliminating short-chain PFAS

The smaller perfluorinated and polyfluorinated alkyl substances (PFAS) are, the more quickly they spread and the more difficult they are to remove. The Helmholtz Centre for Environmental Research (UFZ) has now reported on a new process for separating and destroying perfluorobutanoic acid (PFBA), which is more environmentally friendly and consumes less energy than methods used to date.
PFBA as a typical example
Perfluorobutanoic acid (PFBA) is a typical example of short-chain PFAS that were and still are used by industry, because they are considered a „more harmless“ alternative to long-chain PFAS. But the small molecule size combined with their polarity make them move into soil and water bodies unhinderedly.
PFBA is known to be one of the most detected short-chain PFAS in water bodies, as cited in the publication of this UFZ study. For example, the substance could be detected in drinking water relevant water bodies in USA as well as in Europe, where it was found in the Rhine catchment area and in groundwater reservoirs in France. Particularly high concentrations of about were found in chinese surface waters.
PFBA has only four carbon atoms and ends in a carboxyl group, which strongly attracts water.
“That is why PFBA dissolves readily in water and is highly mobile. It is therefore difficult to remove PFBA from water using conventional methods such as activated carbon adsorption”, says Dr Anett Georgi, UFZ chemist and co-author.
First separate and concentrate the PFAS…
The process developed at the UFZ consists of two electrochemical steps. In the first step, PFAS are separated from water and concentrated by means of electrosorption. In the second step, the molecules are broken down by electro-oxidation. This leaves mainly CO₂ and fluoride (see process diagram below).
Electrosorption takes place in a flow cell on a positively charged electrode made of activated carbon fibre fleece. The fleece attracts the negatively charged PFAS molecules. For desorption, the polarity of the voltage is reversed, causing the molecules to detach from the activated carbon fleece. They are flushed out of the cell with a small volume of water and collected as a concentrate. In this process, the PFAS concentration can be increased by a factor of 40 compared to the treated raw water.
A significant part of the research focused on the flow cell. Among other things, it was necessary to ensure that only the positively charged working electrode selectively adsorbs PFBA, whilst the affinity of the counter electrode must be negligible. Furthermore, no water electrolysis must occur across a wide operating voltage range.
The adsorption electrode is defunctionalised with regard to pure adsorption without charge. This allows it to be fully regenerated in every desorption cycle. This is a significant advantage over conventional activated carbon processes, in which PFAS-loaded activated carbon must be incinerated or regenerated using a complex thermal process.
… then destroy the PFAS molecules
The PFAS contained in the concentrate is broken down in the second stage of the process by electro-oxidation using a boron-doped diamond electrode. The main by-product remaining in the water is fluoride.
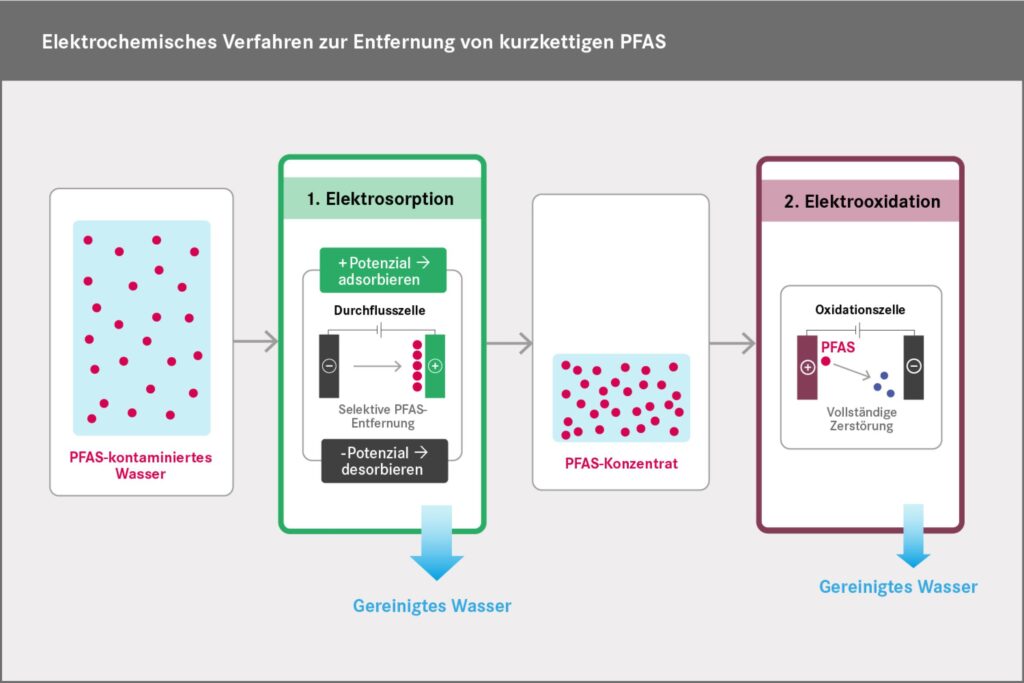
During this process, the PFAS are first separated and concentrated by electrosorption (Step 1) and then destroyed by electrooxidation (Step 2). The main byproducts are CO2 and fluoride.
Image: Susan Walter-Panzner /UFZ (Grafisch)
Applications in PFAS contaminated areas
A patent application has already been filed for the two-stage process. The UFZ researchers see a wide range of potential applications, such as at airports, where groundwater is contaminated with PFAS due to the use of fire-fighting foams.
“In light of the increasingly stringent PFAS limits that operators are required to comply with, there is a need for more efficient removal technologies that are as reliable, environmentally friendly, and affordable as our method. It could complement traditional activated carbon adsorbers in cases of complex PFAS contamination and capture short-chain PFAS”, says Dr Katrin Mackenzie, UFZ chemist and co-author.
This would result in a considerable longer service life of the entire adsorber unit and thus lead to cost savings.
Link to the publication.-
Featured image: photo by Włodek / Pixabay, adaption by myself